Resource recovery, heavy metal removal
- TOP
- Applications of Ion Exchange Resins
- Resource recovery, heavy metal removal
Recovery of metal resources
Looking at the periodic table, there are more than 100 different elements. Broadly speaking, there are 'metal elements' and 'non-metal elements,' and even within what is simply called 'metal,' there are many different types. Iron, aluminum, copper, zinc, tin, and lead are abundant on earth and are called 'base metals.'
On the other hand, other metal elements are classified as 'precious metals,' 'rare metals,' or 'rare earths.'

Precious metals
"Precious metals," as the name suggests, are associated with being valuable, expensive, and shiny, but chemically, they are defined as 'metals that are rare and tend to resist forming compounds.' The elements that meet these criteria are gold (Au), silver (Ag), platinum (Pt), palladium (Pd), rhodium (Rh), iridium (Ir), ruthenium (Ru), and osmium (Os), and these are generally referred to as precious metal elements. In the Olympics, medals are gold, silver, and bronze, but copper is not considered a precious metal.

Rare metal
"Rare metals" do not have a clear definition, but they are generally considered non-ferrous metals that are rare in quantity on Earth or difficult to extract for technical or economic reasons, and whose stable supply is considered important from a policy standpoint (Ministry of Economy, Trade and Industry). They are often used in industry, either to enhance the properties of structural materials, or as functional materials such as electronic materials and magnetic materials. Examples include lithium used in batteries, lightweight yet strong titanium used in aircraft and rockets, chromium and nickel added to iron for stainless steel, and tungsten used in light bulb filaments.

Rare earth
The 17 elements from scandium, yttrium, and lanthanum to lutetium, which are Group 3 elements in the periodic table, are commonly referred to as rare earth elements, or simply “rare earths”. These rare earths are also vital elements underpinning modern industry: neodymium and dysprosium, indispensable for powerful permanent magnets; yttrium, used in powerful solid-state lasers and colour television phosphors; cerium, an abrasive; and terbium, employed in magneto-optical discs. We are largely reliant on imports from China, which accounts for the majority of global production, and the recent issues surrounding these rare earths are still fresh in our minds. However, despite bearing the name “rare”, their abundance on Earth is higher than that of precious metals such as gold and silver, and a certain amount exists on Earth.
When viewed on the periodic table, the following elements correspond to these precious metals, rare metals, and rare earths.
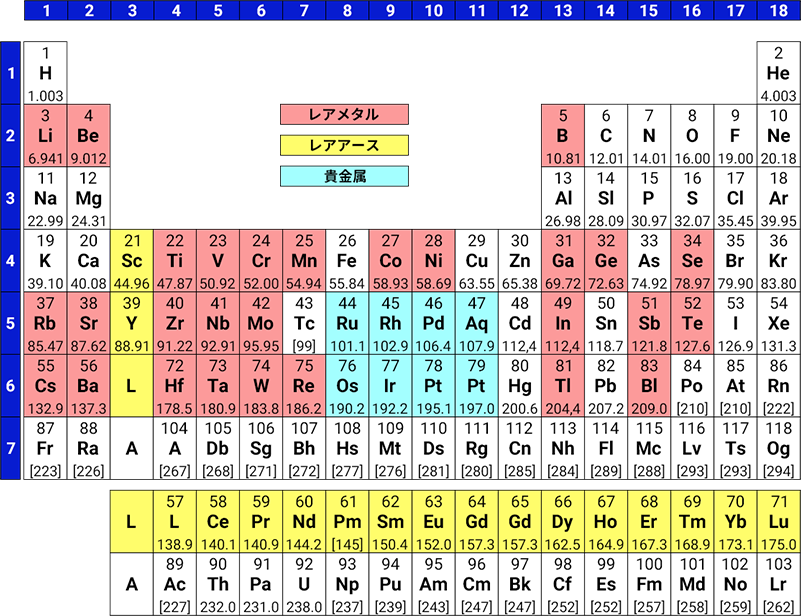 (Pd and Pt may be included as rare metals)
(Pd and Pt may be included as rare metals)
Metal Resources and Heavy Metal Removal
Various methods exist for manufacturing these metal resources, and ion exchange resins or chelating resins may be employed.
Most metals exist as positive ions in water, making them amenable to adsorption and recovery using cation exchange resins. Furthermore, some metals, such as gold and chromium, form complexes with negative ions and exist as negative complex ions. In such cases, adsorption and recovery are possible using anion exchange resins. Additionally, chelating resins are employed when selective recovery of specific metals is desired. Chelating resins possess various functional groups, and by selecting and devising the eluent, it may be possible to recover only specific metals.
Here, precious metals serve as an example. Beyond jewellery worn on the person, precious metals are utilised in a wide variety of applications. It became well known that the medals for the Tokyo 2020 Olympic Games were made using precious metals recovered from waste materials such as PCs and smartphones, referred to as “urban mines”. Examples of precious metals recovered in the industrial sector are shown below. It can be seen that precious metals are used in diverse applications and materials.
【Primary Uses of Precious Metals】
| Industry | Material |
|---|---|
| Semiconductor | Wafers, ICs, LSIs, deposition materials, etc. |
| Surface treatments such as plating | Plating waste liquid, cleaning solution, etc. |
| Photographic materials | Silver halide film, photosensitive materials, etc. |
| Medical field | Dental materials, bone materials, etc. |
| Chemical industry | Catalysts, electrodes, etc. |
| Ornaments | Jewellery, paints, etc. |
| Electronics sector | Mobile phones, smartphones, PCs, televisions, etc. |
In the case of extremely valuable precious metals such as gold, it may be necessary to adsorb them from solutions containing gold using chelating resins or anion exchange resins, and then recover the gold by incinerating the resins.
While various metals are utilised industrially, metal-related pollution problems emerged, particularly during the 1960s (the Shōwa 30s). Those of a certain age will likely be aware that the main issues included the following:
Minamata disease: Mercury
Itai-itai disease: Cadmium
Other toxic diseases: Lead, arsenic, etc.
Minamata disease resulted from organic mercury contained in factory effluent accumulating in fish, causing poisoning symptoms primarily among fishermen who consumed these fish. Itai-itai disease resulted from cadmium contained in drainage from the Kamioka Mine flowing into the Jinzu River basin, causing multiple proximal tubular dysfunction and osteomalacia through crops and drinking water. Other substances harmful to humans exist, and various regulations have been imposed. Measures have been taken in Japan today to ensure people can live safely.
Heavy metals regulated as hazardous substances under health criteria include cadmium, lead, chromium, arsenic, mercury compounds, and selenium. Additionally, heavy metals regulated under environmental quality standards include zinc, copper, soluble iron, soluble manganese, and chromium, while nickel, antimony, molybdenum, and total manganese are designated as items requiring monitoring. As these heavy metals are industrially useful metals, they may be present in typical industrial effluent.
Accordingly, environmental standards and discharge standards (which vary by item but are generally set at approximately ten times the environmental standard) have been established. Should water pollution exceed these standards, countermeasures are required.
Part of the General Discharge Standards established by the Ministry of the Environment is shown below. Permissible limit values are also set for various other items, including hazardous substances and other materials.
【Example of General Effluent Standards】
| Type of substance | Tolerance limit |
|---|---|
| Cadmium and its compounds | 0.03 mg Cd/L |
| Lead and its compounds | 0.1 mg Pb/L |
| Hexavalent chromium compounds | 0.5 mg Cr(VI)/L |
| Arsenic and its compounds | 0.1 mg As/L |
| Mercury and alkyl mercury and other mercury compounds | 0.005 mg Hg/L |
| Alkyl mercury compounds | Not detectable |
| Copper content | 3 mg Cu/L |
| Zinc content | 2 mg Zn/L |
| Soluble iron content | 10 mg Fe/L |
| Soluble manganese content | 10 mg Mn/L |
| Chromium content | 2 mg Cr/L |
Methods for recovering metals present in water, such as factory effluent, include adding alkali to precipitate them as metal hydroxides or adding coagulants to separate and recover them. However, these methods can sometimes struggle to recover trace amounts of metals. In such cases, ion exchange resins or chelating resins are employed. Cation exchange resins adsorb all positive ions, while anion exchange resins adsorb all negative ions, making them widely applicable.
On the other hand, chelating resins are employed when recovering trace amounts of metals from water containing large amounts of sodium ions, for example. Chelating resins possess the characteristic of selectively recovering specific metals from a mixture containing various ions. The type of metal selectively adsorbed differs depending on the type of functional group incorporated into the resin; therefore, selecting the optimal chelating resin is essential. Examples of metals that selectively adsorb to chelating resins are shown below. The optimal resin must be selected based on the properties of the water being treated and the metal to be adsorbed.
【Examples of Chelating Resins and Target Elements】
| Functional Group | Selectivity |
|---|---|
| Iminodiacetic acid | Heavy metals in general. Broadly covering Cu, Hg, Pb, Ni, Zn, Cd, etc. |
| Polyamine | Targeted at mercury and precious metals. |
| Glucamine | Boron selectivity |
| Amidoxime | Recovery of copper and precious metals. |
| Phosphonic acid | Recovery and removal of transition metals |
| Sulphonic acid | Recovery and removal of transition metals |
| Thiourea | Recovery and removal of mercury and precious metals |
| Aminophosphoric acid | Recovery and removal of calcium and metals with low atomic weights |
| Bis-picolylamine | Recovery and removal of copper, nickel, cobalt, etc. |
| Semithiocarbamic acid | Selectively adsorb mercury |
| Thiol | Recovery and removal of silver, copper, lead, cadmium, etc. |
Here, we shall consider the recovery of metals from seawater as one example.
As you know, seawater contains high concentrations of salt (sodium chloride), with 30g present per litre. In contrast, lithium is present at 180mg, uranium at 3.2mg, vanadium at 2mg, nickel at 0.48mg, copper at 0.15mg, and cobalt at 0.0012mg – all trace amounts (Source: Journal of the Japan Society of Marine Science, Vol. 51, No. 5, p. 302 (1997)). Using amidoxime-type chelating resins enables the selective recovery of uranium, vanadium, nickel, and cobalt from seawater. Furthermore, iminodiacetic acid-type chelating resins can also recover copper and cobalt from seawater.
As outlined above, it is evident that technologies employing ion exchange resins and chelating resins for resource recovery and metal removal are widely utilised.
