Pure water systems, ultrapure water systems, water softeners
- TOP
- Applications of Ion Exchange Resins
- Pure water systems, ultrapure water systems, water softeners
Pure water systems and ultrapure water systems
Types and Quality of Water
Our surroundings on Earth are filled with water. It falls as rain, flows into rivers, pours into the sea, evaporates to form clouds (especially as typhoons), and returns to the ground as rain, circulating continuously. Also, when we turn a tap, tap water flows out, which we use for daily drinking and cooking. Factories that produce various goods also use large amounts of water. Water is an indispensable substance for us humans. There are also various types of water.
- Naturally occurring water such as river water, groundwater, and seawater
- Industrial water
- Tap water
- Hard water / Soft water
- Pure water / Distilled water
- Ultrapure water, etc.
Normally, river water is properly treated before being used. There are various types of treatment methods, and the type of water obtained depends on the method used.
| Types of water | Overview of the treatment |
|---|---|
| Industrial water | Water that has been purified to remove trash and turbidity from river water. |
| Tap water | Water that has been purified and sterilized by removing garbage, turbidity, and impurities from river water. |
| Hard water / Soft water | Groundwater suitable for drinking. Depending on the concentration of hardness components, it is classified as 'hard water' or 'soft water'. |
| Pure water / Distilled water | Water that has had impurities removed. |
| Ultrapure water | Water from which impurities have been removed to the utmost extent. |
Industrial water and tap water contain substances other than water, though in amounts less than 1 g per liter. The substances present can be classified into inorganic substances, organic substances, fine particles, and microorganisms. On the other hand, using these waters in industrial fields can sometimes cause problems. In particular, inorganic substances such as calcium and magnesium, referred to as hardness components, and silica (silicic acid) can affect industrial and manufactured products.
Also, regarding tap water, looking at the requirements for tasty water presented by the "Delicious Water Research Committee" established by the Ministry of Health and Welfare at the time, it is clear that various components need to be dissolved in the water. The actual tap water in Tokyo is close to meeting these requirements. Additionally, the hardness of European mineral water is higher than that of Japanese tap water; for example, the Evian website lists its hardness as 304 mg/L.
"Standards for Good-Tasting Water and the Quality of Tap Water in Tokyo"
| Water Quality Parameters | Standards for Good-Tasting Water | Average value for 2013 |
|---|---|---|
| Evaporation Residue | 30~200mg/L | 150 |
| Hardness | 10~100mg/L | 64.6 |
| Free Carbon Dioxide | 3~30mg/L | 2.6 |
| Potassium Permanganate Consumption | 3mg/L or less | 1.0 |
| Odor Intensity | 3 or less | 1 |
| Residual Chlorine | 0.4mg/L or less | 0.4 |
| Water Temperature | Max. 20℃ or less | 16.9 |
(Quoted from the Tokyo Metropolitan Waterworks Bureau website)
"Pure water system" for producing pure water
One of the key industrial products that plays an important role in removing the components contained in water to produce pure water is called 'ion exchange resin.' Table salt is a substance made up of positively charged sodium ions and negatively charged chloride ions, and when dissolved in water, it exists in a dispersed state. Many substances that dissolve in water are in a similar state, and by introducing cation exchange resins that adsorb positive ions and anion exchange resins that adsorb negative ions, the ions in the water are captured, resulting in 'pure water' with few impurities.


A 'pure water system' for producing pure water is composed of the following equipment. Industrial water or tap water is first filtered to remove suspended solids, and then passed through a demineralization tower filled with ion exchange resin to remove impurities from the water, thereby producing 'pure water.' Generally, the '2B3T type' or the 'mixed bed type' is used.
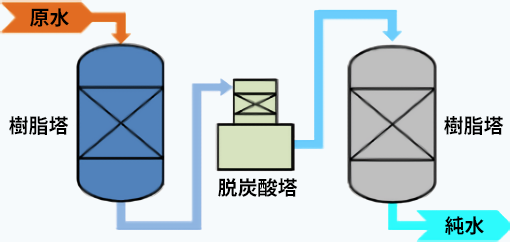
2B3T Type
Mixed-Bed Type
The '2B3T system' produces pure water by filling two demineralization towers with cation and anion resins and passing the water through a decarbonation tower as well. There are also cases where a single tower, called a 'mixed-bed system,' is used with a mixture of cation and anion resins.
There are various configurations for the demineralization towers of pure water systems, and they are selected based on their intended use, required water quality, and cost.
Example Configuration of a Pure Water System
 |
Remove positive ions with a strongly acidic cation resin and remove negative ions with a strongly basic anion resin. |
|---|---|
 |
Remove cations with a strong acid cation resin, remove anions with a type II or weak base anion resin, and further remove anions with a strong base type I anion resin. |
 |
Remove positive ions with a strongly acidic cation resin, remove negative ions with a Type II anion resin, and remove all ions with a mixed-bed resin. |
 |
Remove cations with a strong acid cation resin, further remove anions with Type II, and further remove anions with a strong base Type I anion resin. |
 |
All ions are removed using a mixed bed of strongly acidic cation resin and strongly basic anion resin. |
c: cation, A: anion, M: mixed bed *A decarbonation tower may also be present.
| Device | Features | |
|---|---|---|
| Cation resin tower | Filled with strongly acidic cation resin, it removes positive ions such as sodium, calcium, and magnesium. It also adsorbs rust. | |
| Decarbonation tower | Removal of dissolved carbonic acid in water | |
| Anion resin tower | Removes anions such as chloride ions and silica. An anion resin is filled. | |
| Anion resin tower | Strong Base Type I | It adsorbs -ions over a wide pH range. Silica can also be adsorbed. |
| Strong Base Type II | Its adsorption capacity is slightly lower than Type I, but it is easier to regenerate. Silica can also be adsorbed to some extent. | |
| Weakly basic | Can adsorb -ions in the acidic pH range. Although its adsorption capacity is lower, it is easy to regenerate. It can also adsorb organic substances. | |
| Mixed-bed tower | By using cation resin and anion resin in a mixed state, it is possible to produce high-purity purified water. The equipment has a compact configuration. | |
Ion exchange resins are useful for producing pure water, but they need to be periodically regenerated with chemicals. During this regeneration process, pure water cannot be produced, and there are safety concerns associated with the use of chemicals such as hydrochloric acid and caustic soda.
In recent years, there are cases where pure water production systems using “RO + EDI,” which do not require chemical regeneration, are employed. In this system, water that has been filtered through materials such as activated carbon is passed through a filter called an RO membrane to remove ions and fine particles. After this, a combination of ion exchange resins and ion exchange membranes is used to further remove ions using electrical power in a process called EDI: Electrodeionization, or continuous pure water production with electrical regeneration. Because there is no need to use chemicals, this system has become widely used in recent years.
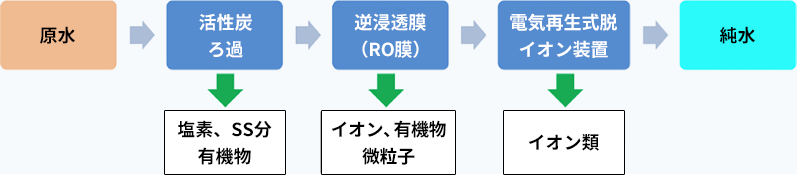
Example configuration of the latest pure water system
This allows the production of 'pure water' with almost no impurities, but there are cases where 'ultrapure water' with even higher purity is used.
It is particularly required for water used in the electronics industry. In semiconductors and liquid crystal components, even trace amounts of impurities can affect product quality. For this reason, components are washed using extremely high-purity water.
In the field of pharmaceuticals, ultrapure water is used not only for cleaning medical devices but also as injection solutions and purified water. The pure water used here is strictly regulated in terms of quality according to the Japanese Pharmacopoeia and similar standards overseas.
In nuclear power plants, electricity is generated by producing steam in the reactor to turn the turbines, and after converting the steam back into water, it is returned to the reactor. At this stage, the water is thoroughly purified and returned as ultrapure water to suppress corrosion of the materials that make up the power generation equipment.
The production of this ultrapure water uses a system that combines multiple treatment facilities. An example of the configuration of an ultrapure water production system used in the electronics industry is shown below.
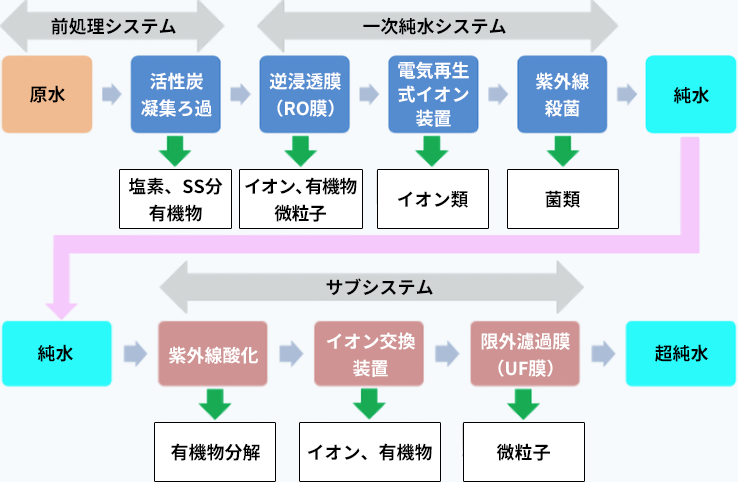
Example configuration of the latest ultrapure water system
First, raw water such as industrial water is treated with pretreatment equipment like activated carbon and coagulation filtration to remove residual chlorine, suspended matter, and organic substances. Next, similar to pure water production systems, an RO membrane and EDI are used to remove ionic components, and ultraviolet light is applied to kill microorganisms, producing pure water. This pure water is then passed through equipment called a subsystem, where organic substances are decomposed by ultraviolet oxidation, ions are removed using ion exchange resins, and finally fine particles are removed by ultrafiltration membranes to produce "ultrapure water," which is used for washing electronic material wafers.
The ion exchange resins used in this subsystem are of high purity, containing very low amounts of impurities such as metals and organic substances, in order to achieve high-quality water (such as our HG series).
Furthermore, there are also challenges in ultrapure water production systems. Ultraviolet lamps are used to kill microorganisms and to oxidatively decompose organic substances. While UV lamps are effective for these purposes, they also produce hydrogen peroxide by decomposing water. This hydrogen peroxide is a troublesome substance for ion exchange resins. In particular, when cation exchange resins come into contact with hydrogen peroxide, they undergo oxidation reactions, leading to a decline in water quality. Therefore, in the final stage of the subsystem, highly oxidation-resistant cation exchange resins are sometimes used (such as in our UL series).
Additionally, pure water and ultrapure water are widely used in universities and research laboratories. In research and chemical analysis, water is analyzed. If the water used for cleaning equipment or performing analytical procedures is not clean, the measurement results will be unstable. Therefore, for example, to determine whether the sodium concentration is higher than 1 ppm, the pure water used must have a concentration lower than this value. Ultrapure water used in electronic materials must meet levels below the detection limit of analytical instruments, so it is naturally necessary for the water to be extremely pure, which requires advanced processing.
Water softener
On the other hand, one type of purified water is "soft water." The difference between soft water and hard water is immediately noticeable when you drink it. This distinct taste comes from the minerals contained in the water, such as calcium and magnesium. Soft water has a lower mineral content compared to hard water, giving it a smooth and easy-to-drink flavor.
The number that indicates the amount of this mineral contained per liter of water is called hardness, and in Japan's general classification, water with a hardness of 100 or less is defined as soft water. Since Japanese water usually has a hardness of around 80, it can be considered quite soft. On the other hand, hard water sold in places like Europe has a heavy texture and a taste that seems to cling to the tongue.
This hardness component is what causes white scale deposits when boiling water in a kettle. In boilers used in factories or public baths to heat water, hardness components can be a hindrance. They are also responsible for the 'water spots' that appear when washing cars.
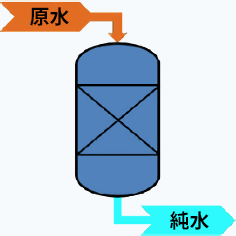
The device used to eliminate these effects is called a "water softener," which uses cation exchange resin. When water containing hardness components like calcium and magnesium passes through sodium-type cation exchange resin, it captures calcium and magnesium and releases sodium. This produces soft water with low hardness. Additionally, regular regeneration can be performed using salt, allowing the resin to be reused. Recently, the use of water softeners has been increasing not only for commercial purposes but also for household use.
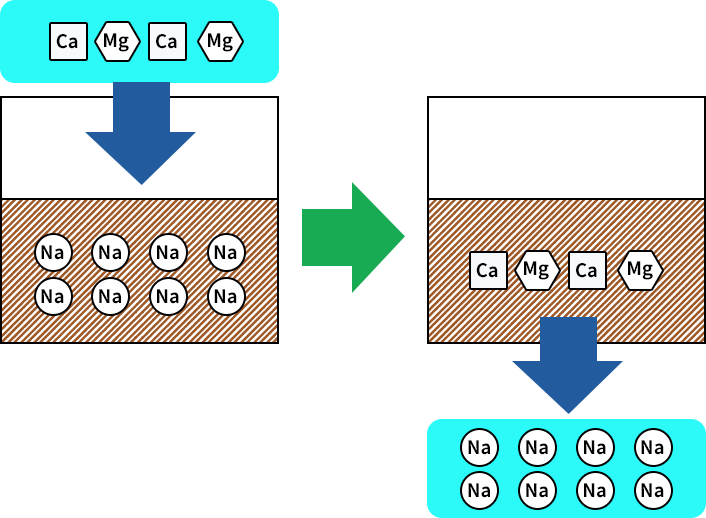
Mechanism of a water softener
