Structure of ion exchange resins (degree of crosslinking, porosity, particle size)
- TOP
- Types of ion exchange resins
- Structure of ion exchange resins (degree of crosslinking, porosity, particle size)
Degree of crosslinking and porosity
Degree of crosslinking
Ion exchange resins absorb water and swell, creating pores within the resin phase. These pores are called micropores, and ions present in water diffuse through these pores to undergo ion exchange. When a large amount of the crosslinking agent DVB is used, the resin has more crosslinking, swells less, and has smaller micropores. Conversely, when less DVB is used, the resin has larger micropores. The proportion of DVB used is referred to as the degree of crosslinking. Generally, a resin with 8% DVB is called a standard crosslinked resin, less than this is called a low-crosslinked resin, and more than this is called a high-crosslinked resin. For water treatment, resins with a crosslinking degree of 8% or 10% are mainly used.
Porosity
Ion exchange resins that are manufactured by polymerizing into a homogeneous gel-like form are called gel-type resins, while resins produced by imparting physical porosity (macropores) to them are called porous-type resins. Macropores are said to help increase the surface area of the resin particles, thereby speeding up ion exchange. Additionally, by adjusting the amount of crosslinking agent, it is possible to regulate porosity and various other properties. The characteristics of ion exchange resins change depending on their structure and degree of crosslinking, each having its own advantages and disadvantages. Understanding these various characteristics is essential for selecting and using the most suitable ion exchange resin; however, this selection is not easy and relies heavily on years of experience.
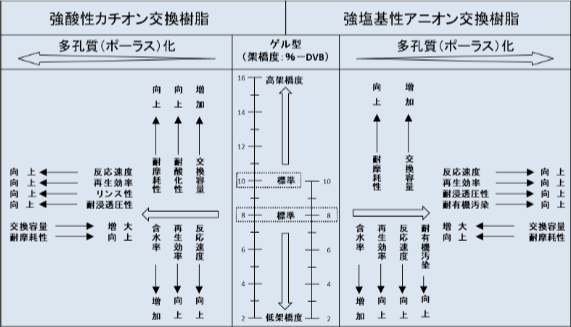
Relationship Between the Structure and Various Properties of Ion Exchange Resins
For example, when looking at the 'exchange capacity,' which indicates the amount of ions that can be adsorbed, ion exchange resins with a high degree of cross-linking or low porosity tend to have higher capacities. On the other hand, when considering the 'reaction speed,' which indicates how easily ions are adsorbed, the trend is the opposite: resins with a low degree of cross-linking or high porosity are more advantageous. Therefore, the choice of ion exchange resin depends on which property is primarily considered.
Particle Size
Ion exchange resins are spherical substances with diameters of about 0.3 to 1.2 mm. As shown in the photo, there are ion exchange resins with a broad Gaussian particle size distribution and those with a uniform particle size distribution where the particle sizes are almost the same. These are used differently depending on their applications, but uniform particle size resins often have advantageous characteristics such as better reactivity and regenerability.

Gaussian distribution ion exchange resin
The particle size distribution is wide.
The packing density is high.

Uniform particle size ion exchange resin
Almost uniform.
Highly reactive and regenerative.
Learn more about the types of ion exchange resins
Structure of Ion Exchange Resins(Crosslinking Degree, Porosity, Particle Size) Cation Resins Anion Resins Chelating Resins High-purity resins
(highly cross-linked resins)
