High-purity resins (highly cross-linked resin)
- TOP
- Types of ion exchange resins
- High-purity resins (highly cross-linked resin)
High-purity ion exchange resins (HG series)

How pure water is produced
Ion exchange resins contain ions that can move within them; cation exchange resins contain positive ions, while anion exchange resins contain negative ions. When these resins come into contact with water, an "ion exchange reaction" occurs by swapping with ions present in the water. If we denote the organic matrix of the ion exchange resin as R, positive ions as A+ and B+, and negative ions as C- and D-, the ion exchange reaction can be represented as follows.
Cation Exchange Resin
A+ + R-B+ ⇆ B+ + R-A+
Anion Exchange Resin
C- + R+D- ⇆ D- + R+C-
By utilizing this ion exchange reaction, when water is passed through a mixed ion exchange resin layer containing both cation exchange resin and anion exchange resin, the cation exchange resin adsorbs cations such as sodium ions and releases H+ ions, while the anion exchange resin adsorbs anions such as chloride ions and releases OH- ions. These H+ and OH- ions combine to form water, resulting in highly purified water (pure water).
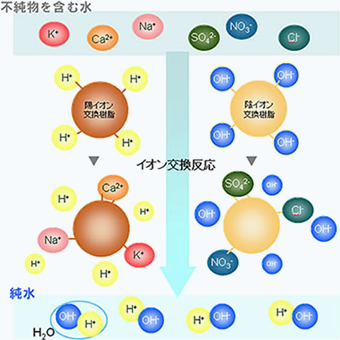
How ultrapure water is made with ion exchange resin
Deterioration of treated water quality
This ion exchange reaction is reversible, allowing both reactions to occur, and this property is utilized for regeneration, making it reusable. However, this characteristic can sometimes cause a deterioration in the quality of the treated water.
New ion exchange resins contain trace amounts of metal impurities originating from the manufacturing process. Although operations such as regeneration and washing are performed to reduce these impurities, they cannot be completely eliminated. As a result, there may be instances where these residual metals are released into the water. When producing ultrapure water or high-purity chemicals, these impurities can have an impact.
Since ion exchange resins are organic substances, any residual organic material remaining from the manufacturing process may not be completely removed during washing and could be released into the water during use. These substances can affect water quality, and in particular, using new resin in equipment such as ultrapure water systems may result in a longer startup time for the equipment.
This ion exchange resin significantly reduces the metal impurities contained in new ion exchange resins, as well as any residual organic substances. It is suitable for the production and purification of pure water, chemicals, organic solvents, and process liquids. We offer a lineup of cation exchange resins, anion exchange resins, and their mixtures. The standard physical property specifications, as well as the startup water quality and metal content when used in pure water systems, are as follows.
"Product Specifications (Guaranteed Values)"
| Item | HG Series | |||
|---|---|---|---|---|
| Mixed-bed Ion-exchange Resin XSM-N411 HG | Cation Exchange Resin | Anion Exchange Resin | ||
| Cation Exchange Resin | Anion Exchange Resin | XSC-1415 HG | XSA-2415 HG | |
| Total Capacity (eq/L-R) | ≥2.1 | ≥1.2 | ≥2.1 | ≥1.2 |
| Average Particle Size(mm) | 0.55〜0.65 | 0.59〜0.69 | 0.55〜0.69 | 0.59〜0.69 |
| Uniformity Coefficient | ≤1.1 | ≤1.1 | ≤1.1 | ≤1.1 |
| TOC (ppb) | ≤30 | ≤50 | ≤30 | |
| Specific Resistance(MΩ・cm) | ≥18.0 | ≥10 | ≥10 | |
| Ionic Form | H+ | OH- | H+ | OH- |
Please scroll horizontally to view the table above.
"Product Information (Reference Values)"
| Item | Cation Exchange Resin | Anion Exchange Resin | |
|---|---|---|---|
| XSC-1415 HG | XSA-2415 HG | ||
| Moisture Retention(%) | 45-50 | 55-65 | |
| Metal content in resin (mg/L-resin) | Al | <10 | <30 |
| Ca、Na | <30 | <10 | |
| Cd、Co、Cr、Cu,K、Li、 Mg、Ni、Pb、Ti、Zn |
<10 | <10 | |
| Fe | <30 | <30 | |
Please scroll horizontally to view the table above.
Additionally, it is effective against organic acids such as citric acid, alcohols such as methanol, and organic solvents such as PGME. The evaluation results for PGME purification are as follows.
| Analysis Items | Raw Liquid | Treated Liquid |
|---|---|---|
| Al | 154 | 0.5 |
| Ca | 146 | 0.2 |
| Co | 118 | < 0.1 |
| Cr | 124 | 2.5 |
| Cu | 129 | < 0.1 |
| Fe | 103 | 0.6 |
| K | 150 | < 0.1 |
| Li | 157 | < 0.1 |
| Mg | 156 | 0.2 |
| Na | 281 | < 0.1 |
| Ni | 119 | < 0.1 |
| Zn | 118 | < 0.1 |
Unit(μg/l)
High Crosslinked Low-Leaching Cation Exchange Resin (UL Series)
Ion exchange resins can remove impurities such as ions present in water and produce clean pure water over a long period of time. However, just as plastic crumbles when exposed to light for a long time, ion exchange resins also gradually degrade with use. In particular, cation exchange resins can undergo oxidative degradation because the metal ions they adsorb and capture act as catalysts. This reaction is said to occur as follows.
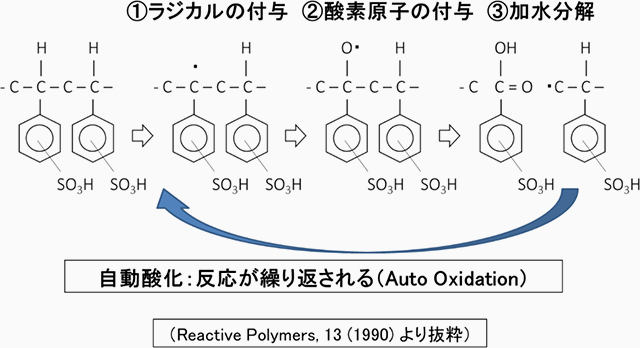
When such reactions occur, the performance of the cation exchange resin deteriorates, leading to the leaching of trace amounts of organic substances. This not only causes a decline in the treated water quality but also becomes a reason for a full replacement of the ion exchange resin.
The solution developed to address this is the "Highly Cross-Linked Low-Leaching Cation Exchange Resin: UL Series." This cation exchange resin, by increasing the degree of cross-linking compared to conventional ones, possesses the following performance characteristics.
- Due to its high degree of crosslinking, it has excellent oxidation resistance and is chemically very stable.
- It has excellent antioxidant properties, making it less likely to cause water quality deterioration due to the leaching of organic substances.
- Because it has low elution of high-molecular-weight organic substances, it is less likely to cause anion resin contamination and has the characteristic of being less prone to water quality deterioration caused by a decline in anion resin performance.
- Compared to standard cation resins, it has a higher exchange capacity, which can be expected to increase water yield.
- It has excellent durability, offering high strength and withstanding long-term use.
- From the above perspective, improvements in water quality, extension of resin lifespan, and increases in water collection capacity can be expected.
The results of evaluating the elution behavior of organic matter are shown below. It was found that high cross-linked, low-elution cation exchange resins release less organic matter than the general-purpose resins used in demineralization units.
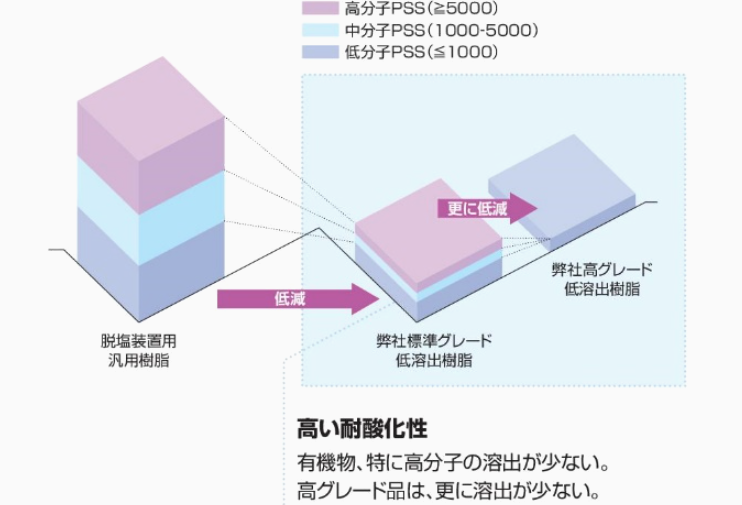
*High grade low-elution resin is a prototype
In addition, the results of the evaluation of ion exchange capacity are shown below. It was found that the high-crosslinking, low-elution cation exchange resin has an exchange capacity approximately 20% higher than that of the general-purpose resin used in demineralization units.
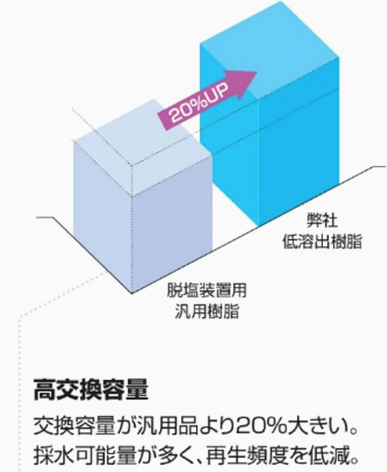
From these perspectives, applying the 'high-crosslinked low-elution cation exchange resin: UL series' can be expected to maintain good water quality while extending the resin's lifespan. Examples of the physical properties of this high-crosslinked cation resin are shown below.
| Item | Standard Grade | High Grade (prototype) | |
|---|---|---|---|
| Ionic Form | Na、H | Na、H | |
| Basic Properties | Total Capacity (eq/L-R) | ≧2.5 | ≧2.5 |
| Moisture Retention(%-Na type) | 28-34 | 28-34 | |
| Whole Bead(%) | ≧95 | ≧98 | |
| No Crack Rate(%) | - | ≧95 | |
| Shipping Gravity(g/ml) | 0.85-0.95 | 0.85-0.95 | |
| Specific Gravity(g/ml) | 1.30-1.40 | 1.30-1.40 | |
| Particle Size | Particle Size 0.315-1.25mm (%) | ≧95 | ≧95 |
| Uniformity Coefficient | ≦1.6 | ≦1.4 | |
| Effective Size(mm) | 0.40-0.70 | 0.40-0.70 | |
| Crushing Strength | Average Strength(g/bead) | - | ≧350 |
| <200g /bead(%) | - | ≦5 | |
| Oxidation Resistance | Low molecular weight PPS(mg/L-R) | - | Low molecular weight PPS(mg/L-R) |
| <200g /bead(%) | - | ≦5 | |
| Oxidation Resistance | Low molecular weight PPS(mg/L-R) | - | ≦300 |
| High molecular weight PPS(mg/L-R) | - | ≦5 | |
| Repeated Regeneration Strength | Crushing & crack rate after 100 cycles(%) | - | ≦2 |
Please scroll horizontally to view the table above.
The high cross-linked cation resin is expected to be used in applications such as the following.
- Condensate demineralization and pure water production equipment for thermal and nuclear power plants
- Pure Water (Make-up Water) Production Equipment for Boiler Feed
- Final-stage polisher for ultrapure water production equipment, such as semiconductor manufacturing equipment
- Pure water system that uses ozone, hydrogen peroxide, or ultraviolet irradiation equipment
- Devices for removing metals from liquids containing oxidizing substances, etc.
Of course, even when used with standard pure water systems, it is effective for maintaining high water purity and prolonging lifespan.
Learn more about the types of ion exchange resins
Structure of Ion Exchange Resins(Crosslinking Degree, Porosity, Particle Size) Cation Resins Anion Resins Chelating Resins High-purity resins
(highly cross-linked resins)
