Chelating resins
- TOP
- Types of ion exchange resins
- Chelating resins
Characteristics of chelating resins
In addition to cation exchange resins and anion exchange resins, there are chelating resins that have functional groups introduced to form chelates with metal ions.
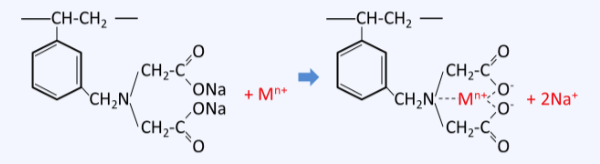
Structure of Chelating Resin and Examples of Reactions with Metal Ions
Chelating resins form very strong complexes with specific metal ions, making them widely used, especially in environmental applications. In particular, chelating resins are used for treating wastewater containing heavy metals such as cadmium, lead, copper, and zinc, which are regulated by discharge standards, and their characteristics are as follows.
- It has selective adsorption for metal ions and can selectively remove and recover specific metal ions.
- It is used for purifying brine and plating solutions, and for removing harmful heavy metals (such as mercury, copper, zinc, and cadmium).
- It is also used for recovering precious metals and valuable metals such as gold, platinum, and palladium.
Functional groups that form chelates contain two or more electron-donating elements such as N, S, O, and P, and include N-O, S-N, N-N, O-O, and P-N types. Representative chelating resins include those with iminodiacetic acid groups, among others.
| Functional Group | Structure | Functional Group | Structure |
|---|---|---|---|
| Iminodiacetic acid | 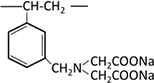 |
Thiol | 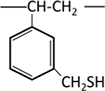 |
| Polyamine | 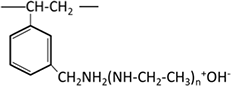 |
Sulfonic acid | 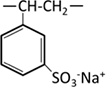 |
| Primary amine | 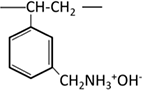 |
Phosphonic acid | 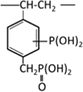 |
| Methylglucamine | 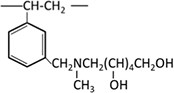 |
Aminophosphonic acid (phosphonate group) | 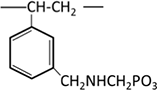 |
| Amidoxime | 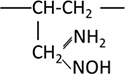 |
Bispicolylamine | 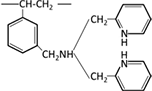 |
| Isothiouronium (thiourea) | 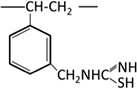 |
Semithiocarbamic acid | 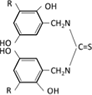 |
| Functional Group | Structure |
|---|---|
| Iminodiacetic acid |  |
| Thiol |  |
| Polyamine | 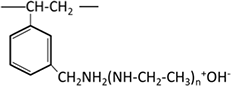 |
| Sulfonic acid |  |
| Primary amine |  |
| Phosphonic acid |  |
| Methylglucamine |  |
| Aminophosphonic acid (phosphonate group) |  |
| Amidoxime |  |
| Bispicolylamine | 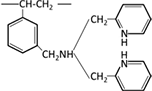 |
| Isothiouronium (thiourea) |  |
| Semithiocarbamic acid |  |
High selectivity of chelating resin
Chelate resins have a much higher selectivity for specific metal ions compared to cation or anion resins. For example, even in aqueous solutions such as saturated sodium chloride solutions or highly concentrated acidic solutions, they can form complexes with and adsorb heavy metals (copper, mercury, lead, etc.) and alkaline earth metals (calcium, magnesium, strontium, etc.), and they hardly release them again unless the pH changes.
Since the selectivity for metal ions varies depending on the type of chelate resin, it is necessary to select the appropriate chelate resin based on the target metal species. An example is shown below.
Iminodiacetic Acid
Hg2+ > Cu2+ > Pb2+ > Ni2+ > Zn2+ > Cd2+ > Co2+ > Fe2+ > Be2+ > Mn2+ > Ca2+ > Mg2+ > Sr2+ >> Na+, K+
Isothiouronium
High selectivity for precious metals such as mercury, gold, platinum, and palladium
Methylglucamine
High selectivity for boric acid
Precautions When Using Chelate Resins
Metal ions adsorbed onto chelating resins can be eluted, recovered, and reused using strong acids such as hydrochloric acid or sulfuric acid, just like with cation exchange resins. However, it is necessary to determine the optimal type and concentration of chemicals depending on the chelating resin used and the species of metal ions to be recovered. In many cases, these settings are largely based on years of practical experience.
- The ease with which metals are adsorbed varies depending on the type of chelate.
- The order of ease of adsorption varies depending on the environment, such as the liquid composition and pH.
- It is necessary to select the type and concentration of chemicals, such as acids, used for regeneration. In particular, for metal species that are highly prone to adsorption, desorption may be difficult.
- The adsorption of metal ions can cause significant volume changes (swelling).
- When eluting adsorbed metal ions, there may be cases where metal oxide deposition occurs.
Learn more about the types of ion exchange resins
Structure of Ion Exchange Resins(Crosslinking Degree, Porosity, Particle Size) Cation Resins Anion Resins Chelating Resins High-purity resins
(highly cross-linked resins)
