Cation resins
- TOP
- Types of ion exchange resins
- Cation resins
Characteristics of cation resins
Cation exchange resin (cationic resin) contains fixed negatively charged ions internally, with positively charged ions present to electrically neutralize them. Since these positive ions can be exchanged with other ions, it is widely used to adsorb cations present in water.
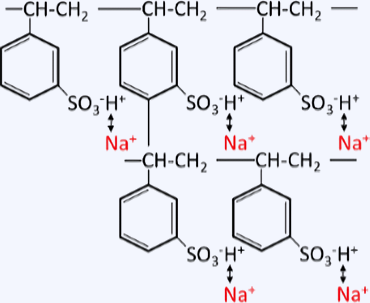
Ion exchange principle of cation exchange resin
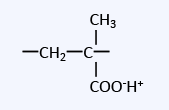
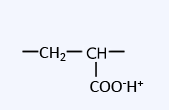
Cation exchange resins can be broadly classified into two types: strong acid cation exchange resins with sulfonic acid groups as functional groups, and weak acid cation exchange resins with carboxylic acid groups.
| Ion-Exchange Resin | Functional Group | Structure |
|---|---|---|
| Strong acid cation exchange resin | Sulfonic acid group | 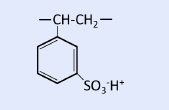 |
| Weak acid cation exchange resin | Carboxylic acid group (Methacrylic type) |
 |
| Carboxylic acid group (Acrylic type) |
 |
Highly versatile strong acid cation exchange resin
-
Strongly acidic cation exchange resins can be used over a wide pH range and can adsorb any cations, making them highly versatile for applications ranging from pure water production to metal recovery from wastewater. This strongly acidic cation exchange resin can be represented as R-SO3H when the polystyrene part is denoted by R, and because the sulfonic acid group is a strong acid group, it dissociates in water regardless of the pH as follows.
R-SO3H ⇆ R-SO3-(fixed ion) + H+(mobile ion) -
An ion exchange reaction occurs when movable ions are exchanged with other positive ions, and if the electrolyte being exchanged is a basic or weakly acidic salt, the ion exchange occurs as follows. Since this reaction is a neutralization reaction, it proceeds easily and can be exchanged even in a batch-type process.
R-SO3H + NaOH → R-SO3Na + H2O2R-SO3H + Ca(HCO3)2 →(R-SO3)2Ca + H2CO3 -
When the exchanged electrolyte is a neutral salt, the exchange reaction produces a mineral acid, which can undergo the reverse reaction, resulting in an exchange equilibrium. Performing this exchange in a batch process leads to the establishment of the exchange equilibrium, preventing the exchange reaction from being completed.
R-SO3H + NaCl ⇆ R-SO3Na + HCl2R-SO3H + CaCl2 ⇆(R-SO3)2Ca + 2HCl -
Ion exchange resins vary in how easily they exchange ions depending on the type of ion. In general, strong acid resins tend to more readily capture ions with higher charges and ions that are less hydrated (those with larger atomic numbers). This tendency follows the following order.
Strong Acid Cation Resins:Al3+>Ca2+>Cu2+>Zn2+>Mg2+>K+>NH4+>Na+>H+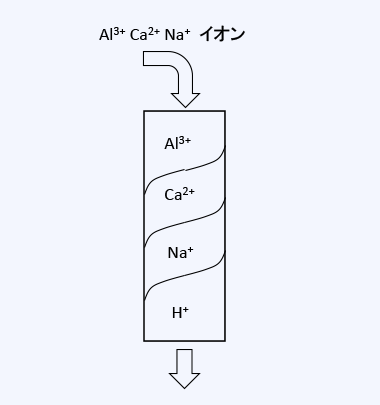
Example of ion separation by selectivity
For example, when water containing Al3+, Ca2+, and Na+ ions is passed through an H-type cation exchange resin, as shown in the figure, an adsorption zone for Al3+, which is the most easily adsorbed, forms at the top of the resin layer, followed by a Ca2+ adsorption zone, and then a Na+ adsorption zone in the lower layer, while the bottom contains H-type resin that does not participate in ion exchange. This characteristic is utilized to adsorb ions present in the water.
On the other hand, since ion exchange reactions are reversible, this characteristic is utilized in the 'regeneration' process. This involves reversing the adsorbed ions to their original state by passing a large amount of high-concentration chemicals through the cation exchange resin at the point when the ion exchange reaction has progressed. In general water treatment applications, Na ions or Ca ions, for example, are loaded onto a cation exchange resin that has adsorbed H ions. By flowing mineral acids such as hydrochloric acid or sulfuric acid, which contain a high concentration of H ions, it becomes possible to return the adsorbed ions to H ions, allowing the resin to be used again.
-
The basic principle of ion exchange reactions is the same for weakly acidic cation exchange resins as it is for strongly acidic cation exchange resins, but they have the following characteristics.
- Has a high exchange capacity per unit volume
- Easy to restore exchange capacity through regeneration
- Cannot be used in acidic environments
- The ion selectivity is low
Making use of these characteristics, weakly acidic cation exchange resins are used in limited applications, such as the removal of hardness components (calcium and magnesium) from groundwater and tap water.
Learn more about the types of ion exchange resins
Structure of Ion Exchange Resins(Crosslinking Degree, Porosity, Particle Size) Cation Resins Anion Resins Chelating Resins High-purity resins
(highly cross-linked resins)
