Anion resin
- TOP
- Types of ion exchange resins
- Anion resin
Characteristics of anion resins
Anion exchange resin (also called anion exchange resin) contains fixed positively charged ions inside, which are electrically neutralized by negatively charged ions. Since these negative ions can be exchanged with other ions, anion exchange resins are widely used to adsorb negative ions present in water.
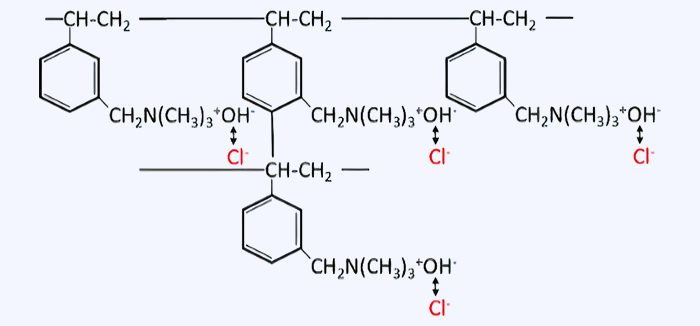
Ion exchange principle of anion exchange resin
Anion exchange resins can be broadly divided into two types: strongly basic anion exchange resins with quaternary amine functional groups, and weakly basic anion exchange resins with primary to tertiary amine functional groups.
| Ion-Exchange Resin | Functional Group | Structure |
|---|---|---|
| Strongly basic anion exchange resin | Quaternary amine Trimethylammonium group (Type I) | 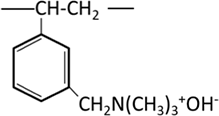 |
| Quaternary amine Dimethylethanolammonium group (Type II) | 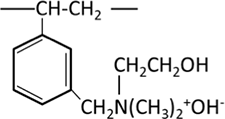 |
|
| Weakly basic anion exchange resin | Tertiary amine (polyamine) | 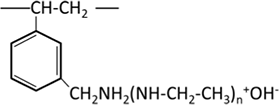 |
| Tertiary amine (dimethylamine) | 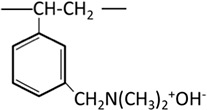 |
|
| Primary amine | 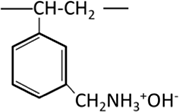 |
General applications of anion resin
-
Strongly basic anion exchange resins can be used over a wide pH range and can adsorb any type of anion, making them highly versatile and commonly used. This strongly basic anion exchange resin can be represented as R-NOH, where R represents the polystyrene part and N represents the quaternary ammonium group. Since the quaternary ammonium group is a strong base, it dissociates in water regardless of the pH as follows.
R-NOH ⇆ R-N+(fixed ion) + OH-(mobile ion) -
An ion exchange reaction occurs when movable ions are exchanged with other positive ions, and if the electrolyte being exchanged is a basic or weakly acidic salt, the ion exchange occurs as follows. Since this reaction is a neutralization reaction, it proceeds easily and can be exchanged even in a batch-type process.
R-NOH + HCl → R-NCl + H2OR-NOH + NH4Cl → R-NCl + NH4OH -
When the exchanged electrolyte is a neutral salt, alkali is produced through the exchange reaction, which causes the reverse reaction to occur, leading to the establishment of an exchange equilibrium. If this exchange is carried out in a batch system, an exchange equilibrium is reached, and the exchange reaction cannot be completed.
R-NOH + NaCl ⇆ R-NCl + NaOH -
Strongly basic anion exchange resins are classified into two types: Type I, which has a trimethylammonium group R-N+(CH3)3OH-, and Type II, which has a dimethylethanolammonium group R-N+(CH3)2CH2CH2OH-. Both are strongly basic, but Type II has slightly lower basicity and inferior exchange capacity for weak acid ions, while it is easier to regenerate. They are used differently depending on the application. Like cation exchange resins, the ease or difficulty of exchange in anion exchange resins also depends on the type of ions.
Strongly Basic Anion Resin: SO42->I->NO3->Br->Cl->OH-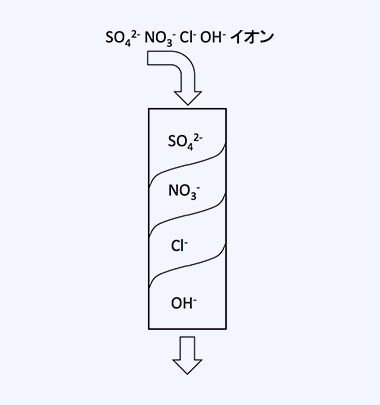
Example of ion separation by selectivity
For example, when water containing SO42- and Cl- ions is passed through an OH-type anion exchange resin, an adsorption zone for SO42-, which is most easily adsorbed, forms at the top of the resin layer, followed by an adsorption zone for Cl-, and at the bottom, there remains OH-type resin that does not participate in ion exchange. Utilizing this property allows the ions present in the water to be adsorbed. In addition, for water treatment, the adsorption of mineral acids can be achieved with either strong or weak base anion exchange resins, but the removal of silicate (silica), which may be present as a target contaminant, is only possible with strong base anion exchange resins.
Since ion exchange reactions in anion exchange resins are reversible, this property is utilized to perform a 'regeneration' process. This involves restoring the adsorbed ion species to their original state by passing a large volume of a high-concentration chemical through the resin once the ion exchange reaction has progressed. When used in general water treatment, ions such as Cl ions are loaded onto the anion exchange resin on which OH ions are adsorbed. By flowing an alkaline solution such as sodium hydroxide, which contains a high concentration of OH ions, over the resin, it is possible to return the adsorbed ions to OH ions, allowing the resin to be used again.
-
Weakly basic anion exchange resins operate on the same basic ion exchange principles as strongly basic anion exchange resins, but they have the following characteristics.
- Has a high exchange capacity per unit volume
- Easy to restore exchange capacity through regeneration
- Cannot be used in alkaline environments
- The ion selectivity is low
Taking advantage of these characteristics, it is used in limited applications such as water treatment and the removal of organic acids. Additionally, because it contains nitrogen atoms with electron pairs as functional groups and can form complexes with metals, weakly basic anion exchange resins are sometimes used as chelating resins for metal recovery.
Learn more about the types of ion exchange resins
Structure of Ion Exchange Resins(Crosslinking Degree, Porosity, Particle Size) Cation Resins Anion Resins Chelating Resins High-purity resins
(highly cross-linked resins)
