Types of ion exchange resins (cation, anion, chelate)
- TOP
- Types of ion exchange resins (cation, anion, chelate)
Cation resin and anion resin
Ion exchange resin is an organic material (synthetic resin) that has the ability to adsorb ions present in water. It is a spherical substance with a diameter of about 0.3 to 1.2 mm to allow water to pass through easily. When an electrolyte dissolves in water, it dissociates into positive ions (cations) and negative ions (anions). The ion exchange resin is created by chemically fixing either the positive or negative ions onto a polymer. The ions bound to the polymer are called fixed ions, and they exist electrically neutralized with ions of the opposite charge, allowing these ions to be exchanged with other ions. This is the ion exchange phenomenon.
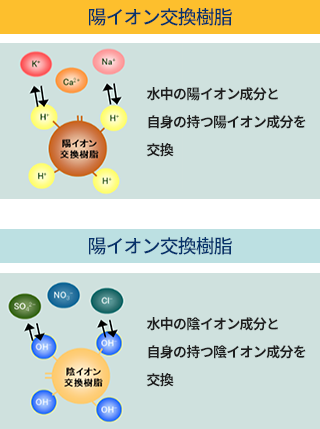
Specifically, cation exchange resins, which have negatively charged sulfonic acid groups as fixed ions, contain positively charged H ions that can undergo ion exchange reactions with Na ions present in water. Similarly, anion exchange resins, which have positively charged trimethylamino groups, contain negatively charged OH ions that can undergo ion exchange reactions with Cl ions present in water. This is the chemical principle behind ion exchange reactions using ion exchange resins.
Ion exchange resins are mainly made from copolymers polymerized from styrene and divinylbenzene (DVB). DVB is used to crosslink polystyrene, making it physically and chemically stable. The resulting copolymer (called crosslinked polystyrene) is hydrophobic, but when functional groups are introduced into it, it becomes hydrophilic, absorbing water and swelling.
For example, when a copolymer is heated in concentrated sulfuric acid, sulfonic acid groups (-SO3) can be attached to produce a strongly acidic cation exchange resin (commonly called a cation exchange resin).
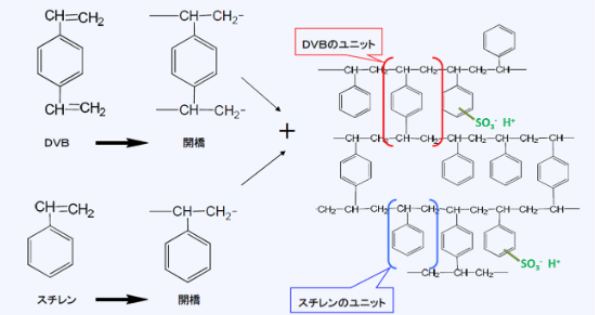
Chemical structure of cation exchange resin
Additionally, through a two-step reaction involving chloromethylation and amination, trimethylamino groups (-N(CH3)3) can be attached to produce an anion exchange resin (commonly called an anion exchange resin).
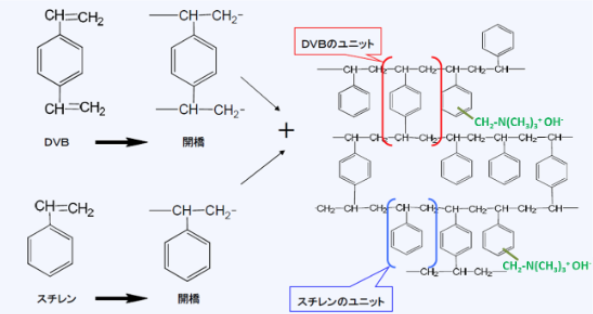
Chemical structure of anion exchange resins
Chelating resins
There is a type of ion exchange resin called chelating resin, which introduces functional groups that form complexes with metal ions. When the ligands contain multiple electron-donating elements such as nitrogen, oxygen, ions, or phosphorus, they strongly bind to metal ions and form complexes. By utilizing this property, functional groups capable of forming complexes with metal ions are introduced into ion exchange resins, and because they have specific selectivity for certain metal ions, they are used for special applications such as the recovery of valuable metals.
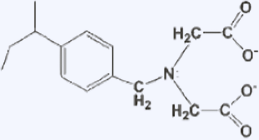
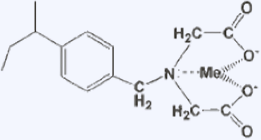
Structure and Reactions of Chelating Resin (Example)
Ion exchange reaction using ion exchange resins
Ion exchange reaction
Ion exchange resins are mainly used for water softening and purification, and generally, cation exchange resins and anion exchange resins are used. In cation exchange resins, H+ ions are present in the resin, and these H+ ions undergo an ion exchange reaction with cations (Na+) in the external solution. In anion exchange resins, OH- ions in the resin exchange with anions (Cl-) in the external solution. Through this process, ions in the water are exchanged with ions in the ion exchange resin. Additionally, once a certain amount of ions have been adsorbed, a "regeneration" process using chemicals is performed to restore the adsorbed ions, allowing the resin to be used repeatedly.
Learn more about the types of ion exchange resins
Structure of Ion Exchange Resins(Crosslinking Degree, Porosity, Particle Size) Cation Resins Anion Resins Chelating Resins High-purity resins
(highly cross-linked resins)
