Applications of Ion Exchange Resins (Their History)
- TOP
- Applications of Ion Exchange Resins (Their History)
Creation of ion exchange resin
By the 1930s, it was discovered that phenol and formaldehyde-based polymers could exchange and adsorb cations and anions, leading to their industrial production as ion exchange resins. By the 1940s, ion exchange resins made from styrene-based polymers, similar to those used today, were being synthesized industrially.
At that time, the world was in the midst of what is known as the Second World War. Ships were powered by steam boilers, and to operate boilers (perhaps better referred to as battleships or transport ships) stably over long periods while mitigating the effects of hardness components, there arose a need for ion exchange resins to purify the water used. This is said to be one of the reasons development progressed. Industrialization research and development was taking place not only in Europe and the United States but also within Japan. Additionally, ion exchange resins were used for the separation of isotopic elements such as uranium.
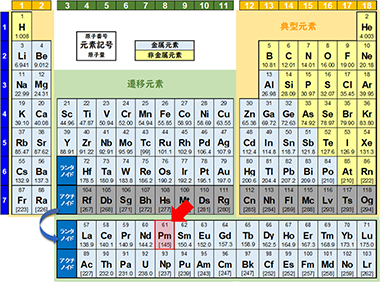
Promethium, atomic number 61
The element name promethium, with atomic number 61, is derived from Prometheus, the god of fire in Greek mythology. Promethium does not occur naturally and is separated from the fission products of uranium using ion exchange resin. Ion exchange was also a technology that played a part in the development of atomic bombs using uranium.
Applications of Ion Exchange Resins
After World War II, along with rapid economic growth, ion exchange resins came to be widely used. They are extensively employed not only for producing pure water, but also for softening hard water, purifying boiler water, and refining substances such as sugar, citrus beverages, and alcohol. They have become a common technique for separating and purifying substances present in liquids. Regarding the purification of boiler water, they are widely used in thermal power plants and nuclear power plants, with massive amounts of ion exchange resin—over 100,000 liters—used in a single power plant. The amount of ion exchange resin used is among the highest in any industry. In industrial applications, they are also used in various other ways.

Semiconductor industry
In the semiconductor industry, such as for memory and CPUs, extremely pure 'ultrapure water' is produced using very clean ion exchange resins with minimal impurities, combined with other technologies. Ultrapure water is an essential technology in the manufacturing of computers and smartphones.
Purification of pharmaceuticals and recovery of precious metals
Furthermore, it is also used in the purification of pharmaceuticals and in the recovery of precious metals from gold ore and urban mines. Here, special ion exchange resins that can selectively recover only pharmaceuticals or precious metals are used. In this way, it can be said that ion exchange resins are essential products in our daily lives.

Scientific equipment
At the 'Super-Kamiokande,' a world-renowned scientific facility in Japan, a large amount of ultrapure water is used.

Super-Kamiokande
There is an abundance of extremely pure groundwater around the Kamioka Mine, but extremely pure water is required for neutrino detection. In particular, substances known to be present in hot springs, such as 'radon' and 'radium,' are serious impurities for neutrino detection, so it is necessary to highly purify them. For this reason, various purification systems, including ion-exchange resins, have been installed. As is well known, this research led to Professors Koshiba and Kajita receiving the Nobel Prize, and ion-exchange resin technology played a key role. Currently, a massive Super-Kamiokande, 8.4 times the size of Super-Kamiokande, is being built, and ultrapure water is also planned to be used there.
(From the Super-Kamiokande official website)
Ion exchange resins commonly used in everyday applications
On the other hand, ion exchange resin technology is also used in familiar, everyday situations.

soft water
It is said that using soft water instead of hard tap water improves soap lather and makes hair silky. Also, in simmered dishes, using soft water can enhance absorption, which is why soft water is considered suitable for Japanese cuisine. A soft water device filled with ion exchange resin is used to produce soft water, so it is sometimes installed in restaurants, beauty salons, and homes for showers, washing machines, and drinking purposes. For car washes, soft water or pure water purified with ion exchange resin may also be used. Soft water is used to prevent hardness minerals from adhering to glass or vehicle bodies, and pure water is used as battery refill fluid.

alcohols
Ion exchange resins are also used in the purification of alcoholic beverages. They are used to remove aldehydes, which cause hangovers and are produced during the fermentation process, as well as to remove coloring components to make the beverage clear and colorless. This helps in providing tasty and clean alcoholic drinks.

Research facility
In facilities such as universities where research is conducted, a pure water system is essential. To analyze the concentration of various substances contained in water, pure water with minimal impurities is required. If tap water is used to wash the glassware needed for analysis, impurities present in the tap water can contaminate it, making it impossible to obtain accurate results. Therefore, pure water, which is clean, is necessary, and ion exchange resins are required to produce this pure water. Research institutions always have pure water production systems that supply clean water, and even more purified water systems known as "ultra-pure water production systems".

"Pure Water" and "Ultrapure Water"
Tap water looks very clean, but it contains substances that are harmless to drink as well as substances necessary for disinfection. If we use a 50-meter pool as an example, it would be like having about a million sugar cubes dissolved in the water (at this amount, it wouldn't taste sweet at all). Water purified with ion exchange resins, known as "pure water," would equate to about one sugar cube in a 50-meter pool. Furthermore, in the case of "ultrapure water" used in facilities like the Super-Kamiokande, it is so clean that it would be like having the equivalent of a single grain of rice in Tokyo Dome-sized water—completely undetectable.
As mentioned above, ion exchange resins are used in a wide range of fields, from households to research institutions like universities, and from food factories to power plants, making them an extremely important product for us.
