What is an ion exchange resin (its true nature)?
- TOP
- What is an ion exchange resin (its true nature)?
Structure and Mechanism of Ion Exchange Resins
Our surroundings are filled with water. It rains, flows into rivers, empties into the sea, evaporates to form clouds (especially as typhoons), and falls again as rain, continuously cycling on Earth.
When we turn on a faucet, tap water comes out, which we use daily for drinking and cooking. Water is an essential substance for humanity.
Water is a substance composed of two hydrogen atoms and one oxygen atom, known as H2O. However, the water we see actually contains various other substances. Seawater is known to contain about 30 grams of salt (sodium chloride) per liter, which can be recognized by its salty taste. On the other hand, even clear tap water contains less than 1 gram per liter of other safe-to-drink impurities. These are generally substances containing calcium and magnesium, commonly referred to as 'hardness components.' European mineral waters typically have high hardness (more of these minerals), while Japanese mineral waters have low hardness (less of these minerals), roughly about one-third of the European level.

Although tap water may look clean, it can pose problems when used industrially. When you boil tap water in a kettle, white limescale eventually forms, which is caused by hardness components in the water, such as calcium and magnesium. In industrial settings, boilers produce large amounts of hot water, and when limescale accumulates, it must be removed. Additionally, seawater contains large amounts of salt, making it undrinkable unless the salt and other components are removed to make it suitable for consumption. Nowadays, if you search online, you can find "pure water" with few impurities being sold, which is used for various purposes such as cleaning contact lenses and refilling batteries.
The industrial product used to remove salt and hardness components contained in this water is called an 'ion exchange resin.' Salt is a substance composed of sodium ions, which carry a positive electric charge, and chloride ions, which carry a negative electric charge, and when dissolved in water, they exist in a separated state. Many substances that dissolve in water exist in the same way, with positive and negative ions present. When a cation exchange resin, which adsorbs positive ions, and an anion exchange resin, which adsorbs negative ions, are introduced, they adsorb the ions in the water, producing 'pure water' with very few impurities.
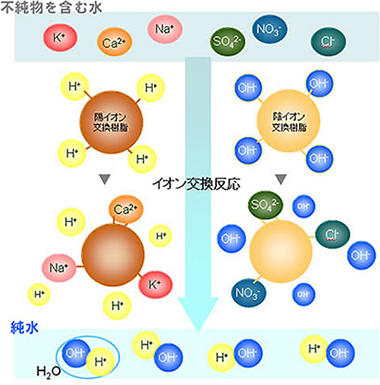
The mechanism by which pure water is produced using ion exchange resin
TIn water, there are positively charged ions such as sodium and negatively charged ions such as chlorine. Cation exchange resins, which absorb positive ions, and anion exchange resins, which absorb negative ions, work respectively, and ultimately, that contains almost no ions: pure water is produced.
Check it out in an even easier-to-understand video!
Ion exchange resin essential for daily life
Ion exchange resins, which are rarely seen in everyday life, are widely used industrially, and we benefit greatly from them. It is used not only for industrial-grade pure water but also for the separation of metals used in electronic materials, the removal of harmful substances from industrial wastewater, and the separation and purification of pharmaceuticals. Moreover, they are used in large quantities for refining sugar, juices, and alcoholic beverages that we consume daily, as well as for purifying water in thermal power plants, making ion exchange resins indispensable in our daily lives.

The industrial use of ion exchange technology began in the mid-19th century with soil improvement techniques. Although the ion exchange phenomenon was not recognized at the time, the technology of 'ion exchange' has been used in the field of agriculture since the Greek era. Farmers utilized it by spreading charcoal, plant ash, and fertilizers on the soil, which induced a type of ion exchange reaction. In Japan as well, records show that fertilizers were used as early as the Heian period. It plays a role in supplying the nitrogen necessary for plant growth to the soil. By the mid-19th century, chemical fertilizers such as phosphate fertilizer and ammonium sulfate began to be synthesized in factories. When applied to soil, ammonium ions, for example, would exchange with calcium ions in the soil, allowing nutrients to be gradually absorbed by plants and function as fertilizer. This demonstrates the use of ion exchange phenomena. In Japan, chemical fertilizers were imported during the Meiji era, and the technology quickly spread.
This natural phenomenon was clarified, and in the early 20th century, 'zeolite,' a composite oxide of the inorganic substances silicon and aluminum, was synthesized. This enabled the effective use of fertilizers and the chemical elucidation of the 'ion exchange reaction.' Subsequently, in the 1930s, ion exchange resins produced through organic synthesis began to be manufactured industrially, leading to the present day.

Appearance of Ion Exchange Resin
Ion exchange resins are organic synthetic substances derived from petroleum, like plastics and chemical fibers. They are very clean, transparent spheres about 0.5 mm in diameter. In appearance, they resemble glassy fish eggs, like cod roe. While not commonly seen in everyday life, they are extremely important chemicals that support various industrial applications.
Learn more about ion exchange resins
Applications of Ion Exchange Resins(Their History)
